Scientists at the National Renewable Energy Laboratory (NREL) have pioneered a method that enables reversible chemistry of magnesium metal in noncorrosive carbonate-based electrolytes. The technology has several potential advantages over lithium-ion batteries, including higher energy density, improved stability and lower cost.
Magnesium batteries can theoretically store almost twice as much energy per volume as lithium-ion batteries. Furthermore, magnesium-metal batteries do not experience the growth of dendrites.
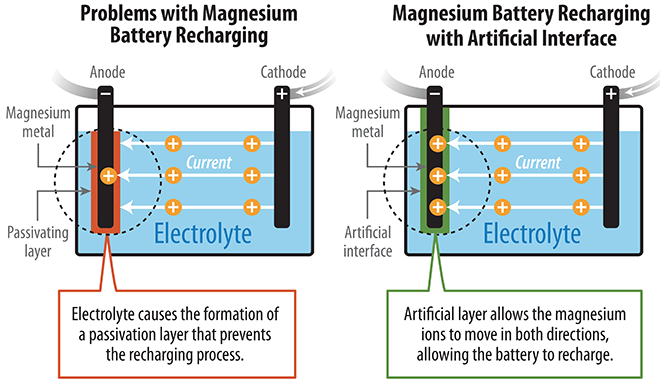
Unfortunately, previous research encountered a major obstacle: the conventional carbonate electrolyte created a barrier on the magnesium surface that prevented the battery from recharging. Magnesium ions could flow in a reverse direction through a highly corrosive liquid electrolyte, but that barred the possibility of a successful high-voltage magnesium battery.
To overcome these roadblocks, the NREL researchers developed an artificial solid-electrolyte interphase from polyacrylonitrile and magnesium-ion salt that protected the surface of the magnesium anode. This protected anode demonstrated markedly improved performance, beyond anything achieved previously. The prototype cell with the protected Mg anode also delivered more energy than the cell without the protection, and continued to do so during repeated cycles.
Source: ChargedEVs